2024-12-16
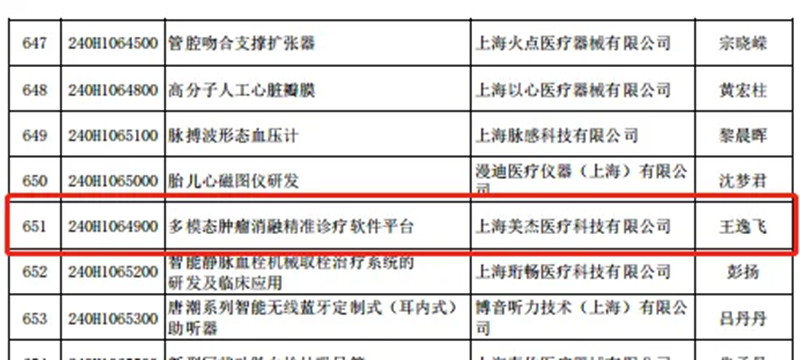
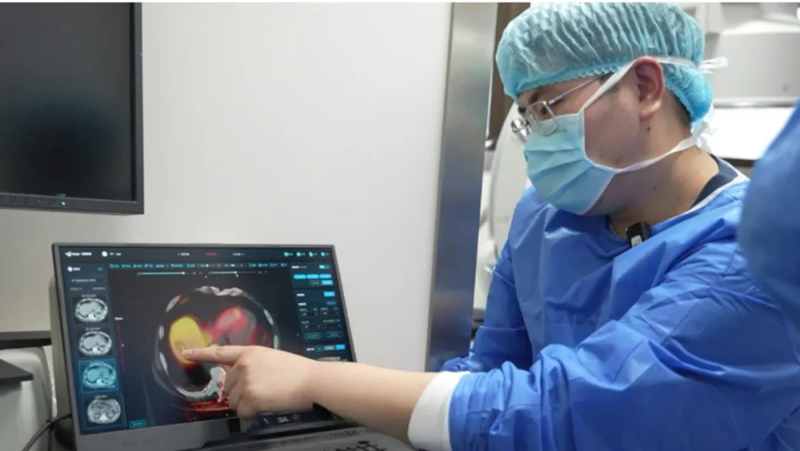
Recently, the Shanghai Science and Technology Commission announced the "List of Approved Projects for the 2024 'Science and Technology Innovation Action Plan' Innovation Fund for Technology‑Based Small and Medium‑Sized Enterprises." The "Multimodal Tumor Ablation Precision Diagnosis and Treatment Software Platform" project, submitted by Shanghai MAaGI Medical Technology Co., Ltd., has been successfully selected.